U.S. CMO/CDMO Market to Reach US$149.4 Bn by 2032 at 8.7% CAGR | Persistence Market Research
The U.S. CMO/CDMO market is rapidly growing, driven by outsourcing, advanced therapies, oncology demand, and emerging biopharma opportunities.
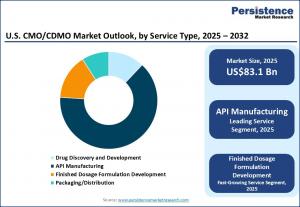
LONDON, UNITED KINGDOM, April 29, 2026 /EINPresswire.com/ -- The U.S. CMO/CDMO market is poised for substantial growth, with a projected valuation of US$83.1 billion in 2025, expected to reach US$149.4 billion by 2032. This trajectory reflects a compound annual growth rate (CAGR) of 8.7% from 2025 to 2032, driven by increasing demand for comprehensive services including API manufacturing, formulation development, packaging, and labeling across pharmaceutical and biopharmaceutical sectors.
Key Industry Highlights
API manufacturing remains the leading service type within the U.S. CMO/CDMO market, largely due to sustained demand for small-molecule APIs and long-term supply contracts with U.S.-based pharmaceutical companies. Oncology emerges as the dominant therapeutic area, propelled by investments in targeted biologics, cell-based therapies, and a growing clinical pipeline. The rapid expansion of emerging biopharma companies, particularly small- and mid-sized firms developing niche and advanced therapies, presents significant growth opportunities for CDMOs. A notable trend is outsourcing manufacturing and development services to CDMOs, which offers U.S. biopharma companies substantial cost advantages by eliminating upfront capital expenditure on GMP-compliant facilities and reducing idle capacity risks.
Download Your Free Sample & Explore Key Insights: https://www.persistencemarketresearch.com/samples/35634
Market Growth Drivers
Outsourcing to CDMOs enables U.S. biopharma firms to achieve cost efficiency and faster commercialization. By partnering with CDMOs, companies gain access to specialized manufacturing capabilities, including API production and sterile injectables, which reduces time-to-clinic and mitigates operational risks. Mid-sized biotech firms increasingly rely on CDMOs to manage production capacity, effectively minimizing cash burn and operational strain. Advanced therapy modalities such as cell, gene, viral vectors, and mRNA further drive market expansion, requiring specialized bioprocessing facilities and expertise. Leading CDMOs have responded by upgrading facilities; for example, Piramal Pharma Solutions invested US$90 million in 2025 to expand sterile injectables and antibody-drug conjugate capabilities at its Lexington and Riverview sites.
Operational Challenges
Despite growth, the U.S. CMO/CDMO market faces rising operational costs and supply chain complexity. The biotech sector requires highly skilled personnel, and shortages, coupled with inflationary input costs for consumables and cold chain logistics, increase expenses. Building and validating new GMP suites is costly and time-intensive, prolonging CAPEX recovery. Supply chain risks, including dependence on limited suppliers and raw material shortages, necessitate higher safety stock levels, tying up capital and challenging CDMOs to balance capacity investments with margin management.
Emerging Biopharma Opportunities
With over 5,000 pharmaceutical and biotech companies in the U.S., demand for CDMO services remains robust, especially in oncology, immunology, and rare diseases. Emerging biopharma firms often lack the infrastructure for end-to-end drug discovery and bioprocessing, driving reliance on CDMOs for integrated services. Increasing clinical trial pipelines and compressed timelines heighten demand for outsourced process development, analytics, cell therapy scale-up, regulatory compliance, and quality assurance. CDMOs provide strategic partnerships that accelerate innovation, minimize capital requirements, and facilitate faster market entry for novel therapies.
Service Type and Therapeutic Area Insights
API manufacturing continues to dominate, offering high-volume, low-risk revenue streams for CDMOs and enabling pharmaceutical companies to leverage advanced manufacturing without fixed asset investments. Oncology remains the leading therapeutic area due to targeted biologics, cell-based therapies, and high R&D spending. CDMOs play a crucial role in guiding the transition from development to commercial manufacturing, meeting urgent clinical timelines.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/35634
Market Trends
Capacity expansion through M&A and facility upgrades is a key trend. Large CDMOs are developing specialized suites and acquiring niche providers to fill technology gaps. For instance, Novo Holdings’ 2024 acquisition of Catalent, Inc., and ESTEVE CDMO’s acquisition of Regis Technologies in 2025 exemplify strategic consolidation. The shift toward end-to-end “one-stop-shop” models allows biopharma firms to streamline regulatory filings, reduce technology transfers, and centralize accountability. U.S. CDMOs are also integrating clinical research functions, offering hybrid CRO-CDMO services that accelerate IND timelines and optimize batch release coordination.
Market Segmentation
By Service
Drug Discovery and Development
API Manufacturing
Finished Dosage Formulation Development
Packaging/Distribution
By Scale of Operation
Preclinical
Clinical
Commercial
By Organization Size
Small-sized
Mid-sized
Large-sized
By Therapeutic Area
Infectious Diseases
Oncology
CNS
Cardiovascular
Respiratory
Others
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/35634
Competitive Landscape and Industry Developments
The U.S. CDMO market is competitive and strategically consolidating. Leading players compete on regulatory track records, technological expertise in ATMPs, ADCs, and viral vector production, along with manufacturing expansion capabilities. Recent developments include Enzene Biosciences’ US$50 million biomanufacturing plant in New Jersey, Lonza’s US$1.2 billion acquisition of Genentech’s Vacaville site, and Fujifilm’s US$1.2 billion investment in North Carolina. Prominent companies in the market include Thermo Fisher Scientific, AGC Biologics, Catalent, Lonza, Piramal Pharma, and Recipharm AB, among others, underscoring a dynamic and evolving market landscape.
The U.S. CMO/CDMO sector is poised for continued expansion, driven by advanced therapies, emerging biopharma growth, and strategic outsourcing trends that collectively reinforce its pivotal role in the pharmaceutical and biopharmaceutical ecosystem.
Read Related Reports:
Hospital Disinfectant Products & Services Market: The global hospital disinfectant products & services market is projected to reach $74.6B by 2033, driven by rising HAIs and strict hygiene regulations.
At-Home Micronutrient Testing Market: The at home micronutrient testing market is set to grow from US$368.4 Mn in 2026 to US$558.4 Mn by 2033, advancing at a steady 6.3% CAGR worldwide.
Persistence Market Research
Persistence Market Research Pvt Ltd
+1 646-878-6329
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.