Active Pharmaceutical Ingredient Market Grows at 5.7% CAGR | Persistence Market Research
Global market analysis of active pharmaceutical ingredients covering trends, growth drivers, regional insights, competition, and opportunities.
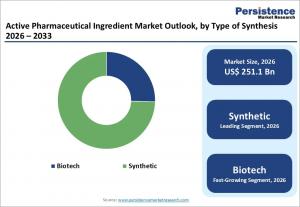
LONDON, UNITED KINGDOM, March 1, 2026 /EINPresswire.com/ -- The global active pharmaceutical ingredient market is projected to grow from US$ 251.1 Bn in 2026 to US$ 370.2 Bn by 2033, registering a CAGR of 5.7% during the forecast period. Historical growth between 2020 and 2025 stood at 4.8%, reflecting steady expansion driven by pharmaceutical production growth and increasing healthcare demand worldwide. Active pharmaceutical ingredients (APIs) are the biologically active components of medicines responsible for therapeutic effects. As global healthcare systems expand and drug consumption rises, API demand continues to increase across both developed and emerging economies.
Download Your Free Sample & Explore Key Insights: https://www.persistencemarketresearch.com/samples/11260
Market Dynamics
Driver: Rising Prevalence of Chronic and Lifestyle-Related Diseases
The increasing global burden of chronic diseases is a primary driver for the API market. Noncommunicable diseases such as cardiovascular disorders, cancer, diabetes, and chronic respiratory illnesses account for approximately 74% of global deaths. In 2021 alone, around 43 million deaths were attributed to such conditions. Diabetes cases have surged dramatically, with about 828 million adults affected globally compared to 200 million in 1990. This expanding patient pool significantly increases long-term medication use, sustaining demand for APIs required in chronic disease treatments. As populations age and urban lifestyles contribute to rising disease prevalence, pharmaceutical production volumes are expected to remain robust, directly supporting API market expansion.
Restraint: High Capital Investment and Operational Costs
API manufacturing involves complex chemical synthesis, stringent quality control, and compliance with Good Manufacturing Practice (GMP) regulations. Establishing production facilities requires significant capital investment in specialized equipment, cleanrooms, and environmental controls. Operational expenses remain high due to regulatory audits, skilled labor requirements, and quality assurance processes. Pharmaceutical companies often face cost of goods sold exceeding 30% of revenue, prompting many to outsource API production to reduce overhead costs. While outsourcing improves cost efficiency, it also creates dependency on external suppliers and global supply chains.
Opportunity: Growth in High-Potency and Specialty APIs
High-potency active pharmaceutical ingredients (HPAPIs) represent a major growth opportunity. Oncology drugs account for over 30% of new drug approvals annually, many of which require highly potent molecules manufactured under specialized containment systems. With more than 1.9 million new cancer cases annually in the United States alone, demand for targeted therapies continues to expand. Advances in biologics, antibody–drug conjugates, and hormone therapies further strengthen demand for HPAPIs. Pharmaceutical firms increasingly collaborate with specialized CDMOs capable of meeting containment, safety, and regulatory standards necessary for these advanced therapies.
Category-wise Analysis
By Type of Synthesis: Synthetic APIs Dominate
Synthetic APIs held approximately 74.5% of the global market share in 2025. The dominance stems from the widespread use of chemically synthesized small-molecule drugs in high-volume therapeutic areas such as cardiovascular, diabetes, anti-infective, and central nervous system treatments. Synthetic APIs account for roughly 70–80% of global API usage due to scalable production processes, cost efficiency, and well-established regulatory pathways. Their affordability and compatibility with generic drug manufacturing further reinforce their leading position.
By Type of Manufacturer: Merchant APIs Lead
Merchant API manufacturers dominate the market as pharmaceutical companies increasingly outsource production. Third-party manufacturers and CDMOs provide scalable, cost-effective solutions without requiring pharmaceutical firms to invest heavily in infrastructure. Outsourcing enables companies to focus on research, development, and commercialization while relying on specialized expertise for API production. This model is particularly prevalent among generic drug manufacturers and small-to-mid-sized pharmaceutical firms, strengthening the merchant segment’s market share.
Get Custom Insights Designed for Your Business: https://www.persistencemarketresearch.com/request-customization/11260
Regional Insights
Asia-Pacific Active Pharmaceutical Ingredient Market Trends
Asia-Pacific accounted for approximately 42.4% of the global market share in 2025, making it the dominant region. Countries such as China and India collectively produce more than 60% of global API volume, driven by large-scale manufacturing infrastructure and competitive production costs. Strong export orientation, government incentives, and regulatory modernization have strengthened the region’s global supply position.
Europe Active Pharmaceutical Ingredient Market Trends
Europe remains strategically important due to its advanced regulatory standards and strong research ecosystem. Regulatory bodies such as the European Medicines Agency enforce stringent GMP standards, ensuring high-quality production. Although some manufacturing has shifted to Asia, Europe continues to lead in complex and high-value APIs, supported by strong pharmaceutical R&D investments.
North America Active Pharmaceutical Ingredient Market Trends
North America is emerging as the fastest-growing region, supported by domestic manufacturing initiatives and supply chain resilience strategies following the COVID-19 pandemic. Companies such as AbbVie Inc. have expanded U.S.-based API production capacity to strengthen domestic supply. Increased investment in biotechnology, rising chronic disease prevalence, and reshoring initiatives are expected to drive sustained growth in the region.
Competitive Landscape
Leading companies focus on scalable production, regulatory compliance, and advanced synthesis technologies. Investments in automation, continuous manufacturing, and process optimization improve efficiency and product quality. Strategic partnerships and capacity expansions ensure consistent API supply for branded, generic, and specialty drug manufacturers worldwide. Prominent players include AbbVie Inc., Bristol-Myers Squibb Company, Merck & Co., Inc., Teva Pharmaceutical Industries Ltd., Sun Pharmaceutical Industries Ltd., Dr. Reddy’s Laboratories Ltd, Aurobindo Pharma, Boehringer Ingelheim International GmbH, Cipla Inc., Mylan N.V., and Albemarle Corporation, all contributing to global API innovation and supply stability.
Checkout Now & Download Complete Market Report: https://www.persistencemarketresearch.com/checkout/11260
Active Pharmaceutical Ingredient Market Segmentation
By Type of Synthesis
Biotech
Synthetic
By Type of Manufacturer
Captive APIs
Merchant APIs
By Application
Cardiovascular Diseases
Oncology
CNS & Neurological Disorders
Pulmonology
Endocrinology
Others
By End User
Pharmaceutical Companies
Biopharmaceutical Companies
CMOs / CDMOs
Others
By Region
North America
Europe
East Asia
South Asia & Oceania
Latin America
Middle East and Africa
Read Related Reports:
Computerized Physician Order Entry (CPOE) Systems Market: Global computerized physician order entry (CPOE) systems market grows from US$2.0 billion in 2025 to US$3.0 billion by 2032 at a 6.3% CAGR.
Picture Archiving and Communication System Market: The global picture archiving and communication system market will hit US$4.4 billion by 2032, at a 6.4% CAGR, driven by rising demand for digital imaging.
Persistence Market Research
Persistence Market Research Pvt Ltd
+1 646-878-6329
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.